A selection of past research is presented below to give an example of research areas our group is interested in.
Canadian and international students and post-docs interested in conducting research with us in similar research areas are welcome to get in touch with me (see how here).
I – STABLE ISOTOPE ANALYSIS of GROUNDWATER CONTAMINANTS
Remediation of groundwater contamination: Monitored Natural Attenuation
A variety of bacteria are able to degrade contaminants under both aerobic and anaerobic conditions. Monitored Natural Attenuation (MNA) is one of possible remediation methods for contaminated groundwater hydrosystems. It relies on intrinsic aquifer microbial processes that lead to contaminant degradation. However, providing strong evidence of in-situ biodegradation of chlorinated aromatic hydrocarbons still remains a challenge.
Compound-Specific stable Isotope Analysis
Field characterization of contaminant concentration or mass decrease over time does not distinguish between degradative (i.e., abiotic or biotic degradation) and non-degradative (e.g., diffusion, adsorption, volatilization) processes that can account for pollutant loss. Conversely, Compound-Specific stable Isotope Analysis (CSIA) has increasingly gained interest as a powerful tool to qualitatively indicate and even quantify in situ biodegradation. This technique is based on the measurement of stable carbon isotope fractionation. Isotopic fractionation originates from differences in reaction rates between the lighter (e.g., 12C) and heavier (e.g., 13C) isotopes of an atom (e.g., C) in a molecule, leading to a shift in the isotopic signature (δ13C) of remaining reactant (i.e., not yet degraded) molecules.
At the University of Toronto, in the Earth Sciences Department, my postdoctoral research with Prof. Barbara Sherwood Lollar focused on the fate of toxic groundwater contaminants. Using CSIA, we evaluated the natural attenuation of these contaminants in situ in the groundwater and at the groundwater/sediment pore water interface.
II – SELENIUM REMOVAL IN CONSTRUCTED WETLANDS
The Salton Sea
In Southern California, the Salton Sea is a terminal lake fed by the New, Whitewater, and Alamo rivers, and that has only one major outflow: evaporation. Due to larger volume of water lost by evaporation than incoming via the rivers, salts, selenium (Se) and other toxic contaminants accumulates in the Salton Sea. The Salton Sea is a major feeding and breeding habitat for many endangered bird species. It is also an important stop-over for migratory birds on the Pacific Flyway. Their survival depends on the preservation of the Salton Sea ecosystem. In the absence of an appropriate intervention, this will result in the destruction of the Salton Sea and a rapid collapse of its precious ecosystem. In response to this major concern, the California Department of Water Resources (DWR) and the California Department of Fish and Game (DFG) have been assigned to develop of a Species Conservation Habitat (SCH) in the Salton Sea, to create a shallow water saline habitat for maintaining fish and bird species dependent upon the Salton Sea.
Selenium contamination
The river water that will feed the SCH contains high levels of contaminants among which selenium (3.18 µgSe/L on average) is one of the most harmful of these pollutants due to its ability to transfer through the food web. Selenium is a naturally occurring element that exhibits an extremely narrow range between essentiality (30 – 85 µgSe/d) and toxicity (> 400 µgSe/d) in human diet. Observed fetus deformities in the Kesterson reservoir have been attributed to Selenium thus urging for finding a solution to treat Selenium-contaminated waters.
Constructed wetlands for selenium removal
Compared to other treatment solutions, constructed wetlands have been identified as the most appropriate technology in many occasions, for presenting the best trade-off between efficiency and cost. Constructed wetlands can remove a very wide range of contaminants including total suspended solids, nitrogen, phosphorus, as well as some metals, pesticides and other micro-organic pollutants. To date, successful experimental constructed wetland studies for Se removal have been conducted at the field scale, e.g., at the Corcoran, Brawley, and Imperial sites in California. However, despite high Se removal rates and evidence of natural Se volatilization, a margin for improving constructed wetland design to increase Se removal rates still exists. Identifying Se species can also give clues to understand which chemical pathways are dominating. It is therefore essential to estimate Se concentrations (ex.: ICP-MS, ICP-OES, AAS) and speciation (ex.: AAS, XAS) for fully evaluating Se reduction and ecotoxicity. Our research in the Plant and Microbial Biology Department at the University of California at Berkeley with Prof. Norman Terry has focused on optimizing the design of constructed wetlands to enhance their natural ability at removing Se.
III – PESTICIDES REMOVAL in FOREST BUFFER and CONSTRUCTED WETLANDS
Dr Passeport’s PhD research at AgroParisTech (with Prof. Yves Coquet) and Irstea (with Dr. Julien Tournebize) focused on the processes that govern the fate of pesticides in an agricultural catchment and two buffer zones: a constructed wetland and a forest buffer, designed to catch and reduce pesticide pollution.
Download Elodie’s PhD Dissertation here (in English). This project was part of the LIFE Environment Project ArtWET and included additional funding from the FIRE (Fédération Ile-de-France de Recherche sur l’Environnement) for work conducted at the INRA (National Institute for Agronomic Research).
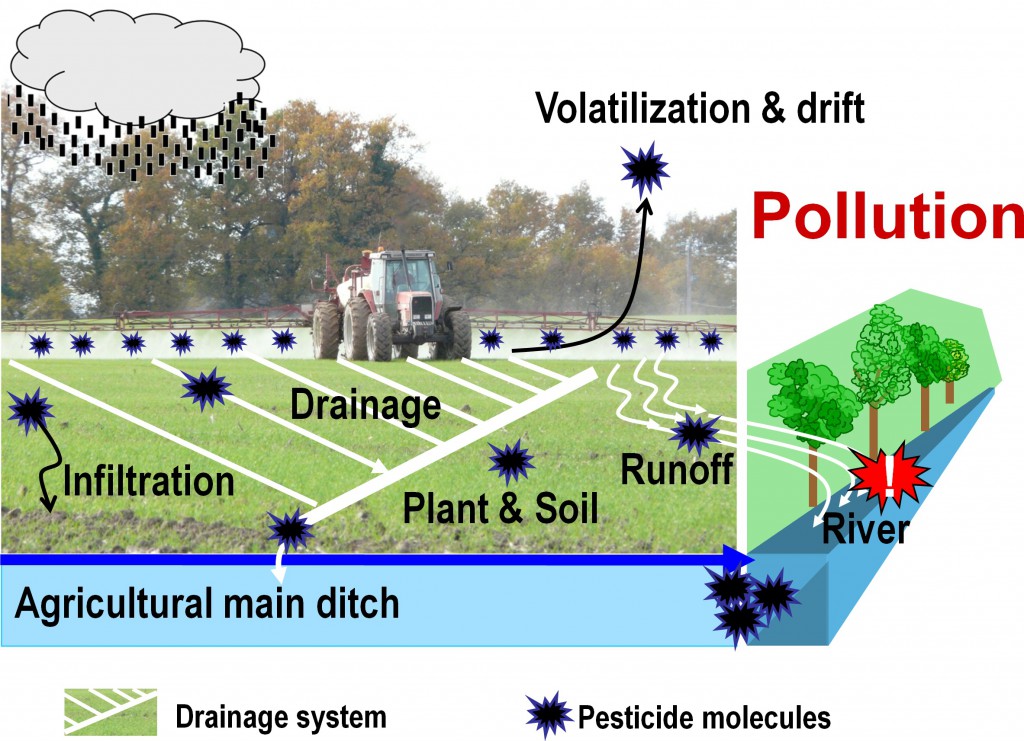
Pesticide transfer in an agricultural watershed
After pesticide application on agricultural watersheds, a portion of applied molecules goes in the air via volatilization or pesticide drift. Some molecules reach targeted organisms, plants, or soil. Finally, after a rainfall event, some pesticide molecules can be transported in the soil profile by infiltration, or transported via tile-drainage or surface runoff towards receiving aquatic ecosystems. According to pesticide applied dose and aquatic ecosystem vulnerability, pesticide use can result in water pollution.
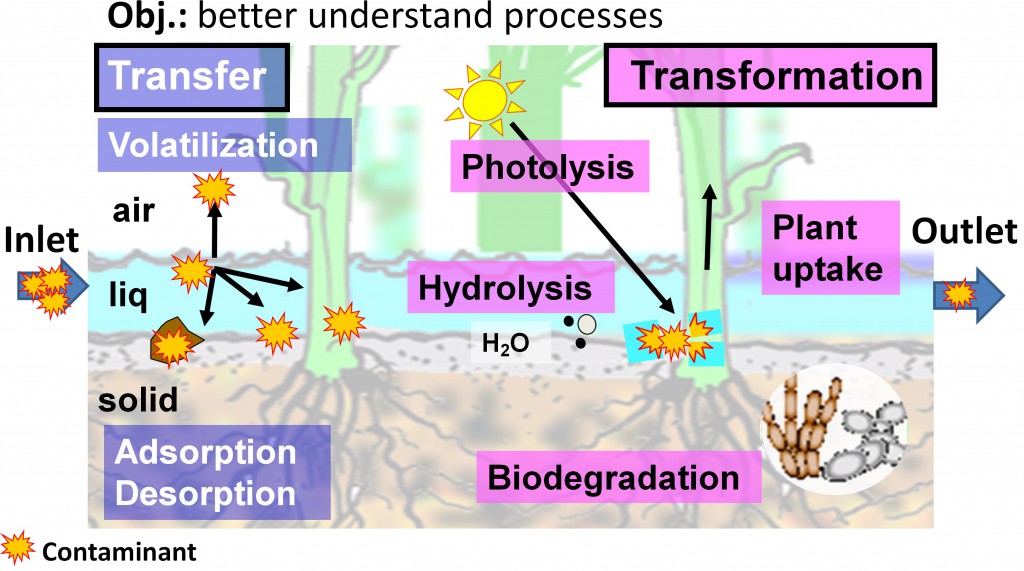
Pesticide dissipation processes in buffer zones
Pesticides can undergo transfer or transformation processes.
When contaminated flows cross buffer zones, some molecules can adsorb onto solid surfaces like plant stems, sediments, suspended particles (a). Particle-laden pesticides can be eliminated from the water column via sedimentation. When water chemistry composition changes, some molecules can desorb and return to the water column (b). Finally, photo-degradation (c), hydrolysis (d) or biodegradation (e) processes can transform the parent pesticide molecule to metabolites, until, possibly, their mineralization.
Buffer zone efficiency for pesticide removal
Buffer zone potential for pesticide mitigation exists. However, a very wide range of pesticide removal efficiencies in constructed wetland was noted. It is extremely delicate to state how much removal can be expected through a given treatment system because such results are pesticide- and site-dependent. However, on average, it is usually admitted that more than 50% of hydrophobic pesticide pollution can be reduced. Forest buffers were part of a very limited number of studies to assess their potential at limiting pesticide pollution but were shown to have potential for pesticide mitigation. Adsorption – desorption and infiltration processes were shown to exert an important role on pollutant elimination from the water column. Microbial degradation can be time-consuming. Consequently, to enhance pesticide degradation, it is crucial to increase water retention time in the treatment systems.